A) Hybridisation is the mixing of atomic orbitals prior to their combining into molecular orbitals
B) \[s{{p}^{2}}\]hybrid orbitals are formed from two p-atomic orbitals and one s-orbital.
C) \[{{d}^{2}}s{{p}^{3}}\]hybrid orbitals are directed towards the comers of a regular octahedron
D) \[s{{p}^{3}}d\]hybrid orbitals are all at \[90{}^\circ \]to one another
E) None of the above
Correct Answer: D
Solution :
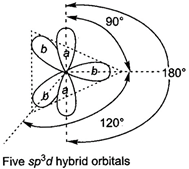 The \[s{{p}^{3}}d\]hybrid orbitals are at \[90{}^\circ ,\text{ }120{}^\circ \]and\[180{}^\circ \]angle.
The \[s{{p}^{3}}d\]hybrid orbitals are at \[90{}^\circ ,\text{ }120{}^\circ \]and\[180{}^\circ \]angle.
You need to login to perform this action.
You will be redirected in
3 sec
