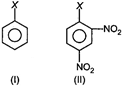
 \[\underset{(\text{III})}{\mathop{{{(C{{H}_{3}})}_{3}}C-X}}\,\] \[\underset{(\text{IV})}{\mathop{{{(C{{H}_{3}})}_{3}}CH-X}}\,\]
\[\underset{(\text{III})}{\mathop{{{(C{{H}_{3}})}_{3}}C-X}}\,\] \[\underset{(\text{IV})}{\mathop{{{(C{{H}_{3}})}_{3}}CH-X}}\,\]
A) \[I<II<IV<III\]
B) \[III<II<IV<I\]
C) \[I<II<III<IV\]
D) \[III<IV<II<I\]
Correct Answer: A
Solution :
AlkyI halides are more reactive than aryl halides. Among alkyl halides, \[3{}^\circ \]alkyl halides (III) are more reactive than \[2{}^\circ \]alkyl halides. (IV) Among aryl halides, aryl halides having electron with dwawing\[-N{{O}_{2}}\]groups at\[o-\]and \[p-\]positions (i.e. II) are more reactive than simple aryl halides (I) thus, the overall reactivity increases in the order. \[I<II<IV<III\]You need to login to perform this action.
You will be redirected in
3 sec
