-
question_answer1) Among the following, the molecule with the lowest dipole moment is
JEE Main Online Paper (Held On 19 May 2012)
A)
\[CHC{{l}_{3}}\]
done
clear
B)
\[C{{H}_{3}}Cl\]
done
clear
C)
\[C{{H}_{2}}C{{l}_{2}}\]
done
clear
D)
\[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
-
question_answer2) If\[{{K}_{sp}}\]of\[Ca{{F}_{2}}\]at \[={{25}^{o}}C\]is\[1.7\times {{10}^{-10}},\]the combination amongst the following which gives a precipitate of \[Ca{{F}_{2}}\] is
JEE Main Online Paper (Held On 19 May 2012)
A)
\[1\times {{10}^{-2}}M\,C{{a}^{2+}}\]and\[1\times {{10}^{-3}}M\,{{F}^{-}}\]
done
clear
B)
\[1\times {{10}^{-4}}M\,C{{a}^{2+}}\]and\[1\times {{10}^{-4}}M\,{{F}^{-}}\]
done
clear
C)
\[1\times {{10}^{-2}}M\,C{{a}^{2+}}\]and\[1\times {{10}^{-5}}M\,{{F}^{-}}\]
done
clear
D)
\[1\times {{10}^{-3}}M\,C{{a}^{2+}}\]and\[1\times {{10}^{-5}}M\,{{F}^{-}}\]
done
clear
View Answer play_arrow
-
question_answer3) The correct order of ligands in the spectro chemical series is
JEE Main Online Paper (Held On 19 May 2012)
A)
\[C{{l}^{-}}>en>C{{N}^{-}}>NC{{S}^{-}}\]
done
clear
B)
\[C{{l}^{-}}>en>NC{{S}^{-}}>C{{l}^{-}}\]
done
clear
C)
\[NC{{S}^{-}}>C{{N}^{-}}>C{{l}^{-}}>en\]
done
clear
D)
\[en>C{{N}^{-}}>C{{l}^{-}}>NC{{S}^{-}}\]
done
clear
View Answer play_arrow
-
question_answer4) The value of\[{{K}_{p}}\]for the equilibrium reaction\[{{N}_{2}}{{O}_{4}}(g)2N{{O}_{2}}(g)\]is 2. The percentage dissociation of \[{{N}_{2}}{{O}_{4}}(g)\]at a pressure of 0.5 atm is
JEE Main Online Paper (Held On 19 May 2012)
A)
25
done
clear
B)
88
done
clear
C)
50
done
clear
D)
71
done
clear
View Answer play_arrow
-
question_answer5)
The order of basicity of the compounds 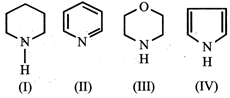 is
JEE Main Online Paper (Held On 19 May 2012)
is
JEE Main Online Paper (Held On 19 May 2012)
A)
\[IV>I>III>II\]
done
clear
B)
\[I>III>II>IV\]
done
clear
C)
\[III>I>IV>II\]
done
clear
D)
\[II>I>III>IV\]
done
clear
View Answer play_arrow
-
question_answer6) Ammonium chloride crystallizes in a body centred cubic lattice with edge length of unit cell of 390 pm. If the size of chloride ion is 180 pm, the size of ammonium ion would be
JEE Main Online Paper (Held On 19 May 2012)
A)
174pm
done
clear
B)
158pm
done
clear
C)
142pm
done
clear
D)
126pm
done
clear
View Answer play_arrow
-
question_answer7) Which pair of elements with the given atomic numbers is expected to have similar properties?
JEE Main Online Paper (Held On 19 May 2012)
A)
40,72
done
clear
B)
20,36
done
clear
C)
10,28
done
clear
D)
11,12
done
clear
View Answer play_arrow
-
question_answer8) Extraction of zinc from zinc blende is achieved By
JEE Main Online Paper (Held On 19 May 2012)
A)
roasting followed by self-reduction
done
clear
B)
electrolytic reduction
done
clear
C)
roasting followed by reduction with carbon
done
clear
D)
roasting followed by reduction with another metal
done
clear
View Answer play_arrow
-
question_answer9) Liquids A and B form an ideal solution. At \[30{}^\circ C\], the total vapour pressure of a Solution containing 1 mol of A and 2 mol of B is 250 mm Hg. The total vapour pressure becomes 300 mm Hg when 1 more mol of A is added to the first solution. The vapour pressures of pure A and B at the same temperature are
JEE Main Online Paper (Held On 19 May 2012)
A)
150,450 mm Hg
done
clear
B)
125,150 mm Hg
done
clear
C)
450,150 mm Hg
done
clear
D)
250,300 mm Hg
done
clear
View Answer play_arrow
-
question_answer10)
The IUPAC name of the following compound is 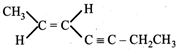 JEE Main Online Paper (Held On 19 May 2012)
JEE Main Online Paper (Held On 19 May 2012)
A)
(E)-2-hepten-4-yne
done
clear
B)
Z)-5-hepten-3-yne
done
clear
C)
(E)-5-hepten-3-yne
done
clear
D)
(Z)-2-hepten-4-yne
done
clear
View Answer play_arrow
-
question_answer11) Which of the following paramagnetic ions would exhibit a magnetic moment (spin only) of the order of 5 BM? (At. Nos. \[Mn=25,Cr=24,V=23,Ti=22\])
JEE Main Online Paper (Held On 19 May 2012)
A)
\[M{{n}^{2+}}\]
done
clear
B)
\[T{{i}^{2+}}\]
done
clear
C)
\[{{V}^{2+}}\]
done
clear
D)
\[C{{r}^{2+}}\]
done
clear
View Answer play_arrow
-
question_answer12) Which one of the following will react most vigorously with water?
JEE Main Online Paper (Held On 19 May 2012)
A)
Li
done
clear
B)
K
done
clear
C)
Rb
done
clear
D)
Na
done
clear
View Answer play_arrow
-
question_answer13) Which of the following has the square planar structure?
JEE Main Online Paper (Held On 19 May 2012)
A)
\[Xe{{F}_{4}}\]
done
clear
B)
\[NH_{4}^{+}\]
done
clear
C)
\[BF_{4}^{-}\]
done
clear
D)
\[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
-
question_answer14) The major product obtained in the photobromination of 2-methyl butane is
JEE Main Online Paper (Held On 19 May 2012)
A)
2-bromo-2-methylbutane
done
clear
B)
1-bromo-2-methylbutane
done
clear
C)
1-bromo-3-methylbutane
done
clear
D)
2-bromo-3-methylbutane
done
clear
View Answer play_arrow
-
question_answer15) If the kinetic energy of an electron is increased four times, the wavelength of the de-Broglie wave associated with it would become
JEE Main Online Paper (Held On 19 May 2012)
A)
one fourth
done
clear
B)
half
done
clear
C)
four times
done
clear
D)
two times
done
clear
View Answer play_arrow
-
question_answer16) When \[C{{O}_{2(g)}}\]is passed over red hot coke it partially gets reduced to \[CO(g).\]Upon passing L of \[C{{O}_{2}}(g)\] over red hot coke, the total volume of the gases increased to 700 mL. The composition of the gaseous mixture at STP is
JEE Main Online Paper (Held On 19 May 2012)
A)
\[C{{O}_{2}}=300mL;CO=400mL\]
done
clear
B)
\[C{{O}_{2}}=0.0mL;CO=700mL\]
done
clear
C)
\[C{{O}_{2}}=200mL;CO=500mL\]
done
clear
D)
\[C{{O}_{2}}=350mL;CO=350mL\]
done
clear
View Answer play_arrow
-
question_answer17)
For a reaction \[A\to \]Products, a plot of \[\log {{t}_{1/2}}\] versus \[\log {{a}_{0}}\]is shown in the figure. If the initial concentration of A is represented by \[{{a}_{0}}\] the order of the reaction is 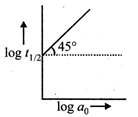 JEE Main Online Paper (Held On 19 May 2012)
JEE Main Online Paper (Held On 19 May 2012)
A)
one
done
clear
B)
zero
done
clear
C)
two
done
clear
D)
three
done
clear
View Answer play_arrow
-
question_answer18) An open vessel at 300 K is heated till 2/5th of the air in it is expelled. Assuming that the volume of the vessel remains constant, the temperature to which the vessel is heated, is
JEE Main Online Paper (Held On 19 May 2012)
A)
1500 K
done
clear
B)
400 K
done
clear
C)
500 K
done
clear
D)
750 K
done
clear
View Answer play_arrow
-
question_answer19) Which of the following is a polyamide?
JEE Main Online Paper (Held On 19 May 2012)
A)
Teflon
done
clear
B)
Orion
done
clear
C)
Nylon
done
clear
D)
Terylene
done
clear
View Answer play_arrow
-
question_answer20) The compound of xenon with zero dipole moment is
JEE Main Online Paper (Held On 19 May 2012)
A)
\[Xe{{O}_{3}}\]
done
clear
B)
\[Xe{{F}_{4}}\]
done
clear
C)
\[XeO{{F}_{4}}\]
done
clear
D)
\[Xe{{O}_{2}}\]
done
clear
View Answer play_arrow
-
question_answer21) The standard potentials of\[A{{g}^{+}}/Ag,H{{g}_{2}}^{2+}/2Hg,\]\[C{{u}^{2+}}/Cu\] and \[M{{g}^{2+}}/Mg\] electrodes are 0.80,0.79, 0.34 and -2.37 V, respectively. An aqueous solution which contains one mole per litre of the salts of each of the four metals is electrolyzed. With increasing voltage, the correct sequence of deposition of the metals at the cathode is
JEE Main Online Paper (Held On 19 May 2012)
A)
\[Ag,Hg,Cu,Mg\]
done
clear
B)
\[Cu,Hg,Ag\]only
done
clear
C)
\[Ag,Hg,Cu\]only
done
clear
D)
\[Mg,Cu,Hg,Ag\]
done
clear
View Answer play_arrow
-
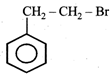
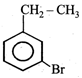
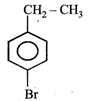
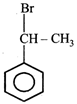
question_answer22) The product of the reaction between ethyl benzene and N-bromosuccinamide is
JEE Main Online Paper (Held On 19 May 2012)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
-
question_answer23) On addition of 1 mL of 10% NaCl solution to 10 mL gold sol in the presence of 0.025 g of starch, the coagulation is just prevented. Starch has gold number
JEE Main Online Paper (Held On 19 May 2012)
A)
2.5
done
clear
B)
25
done
clear
C)
0.25
done
clear
D)
0.025
done
clear
View Answer play_arrow
-
question_answer24) Which of the following is a non-reducing sugar?
JEE Main Online Paper (Held On 19 May 2012)
A)
Lactose
done
clear
B)
Fructose
done
clear
C)
Sucrose
done
clear
D)
Maltose
done
clear
View Answer play_arrow
-
question_answer25) Fog is a colloidal solution of
JEE Main Online Paper (Held On 19 May 2012)
A)
solid particles dispersed in gas
done
clear
B)
solid particles dispersed in a liquid
done
clear
C)
liquid particles dispersed in gas
done
clear
D)
gaseous particles dispersed in a liquid
done
clear
View Answer play_arrow
-
question_answer26) The enthalpy of neutralisation of \[N{{H}_{4}}OH\]with HCl is -51.46 kJ \[\text{mo}{{\text{l}}^{-1}}\] and the enthalpy of neutralisation of NaOH with HCl is -55.90 kJ \[\text{mo}{{\text{l}}^{-1}}\]. The enthalpy of ionisation of\[N{{H}_{4}}OH\] His
JEE Main Online Paper (Held On 19 May 2012)
A)
\[\text{-107}\text{.36}\,\text{kJ}\,\text{mo}{{\text{l}}^{-1}}\]
done
clear
B)
\[\text{-4}\text{.44}\,\text{kJ}\,\text{mo}{{\text{l}}^{-1}}\]
done
clear
C)
\[\text{+107}\text{.36}\,\text{kJ}\,\text{mo}{{\text{l}}^{-1}}\]
done
clear
D)
\[\text{+4}\text{.44}\,\text{kJ}\,\text{mo}{{\text{l}}^{-1}}\]
done
clear
View Answer play_arrow
-
question_answer27) Aspirin can be prepared by the reaction of
JEE Main Online Paper (Held On 19 May 2012)
A)
Salicyldehyde with acetic anhydride in presence of\[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
Salicylic acid with metnanol in presence of\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
Salicylic acid with acetic anhydride in presence of \[{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
Cinnamic acid with acetic anhydride in presence of\[{{H}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
-
question_answer28) Beilstein test is used for the estimation of which one of the following elements?
JEE Main Online Paper (Held On 19 May 2012)
A)
S
done
clear
B)
Cl
done
clear
C)
C and H
done
clear
D)
N
done
clear
View Answer play_arrow
-
question_answer29) Square-planar geometry is shown by
JEE Main Online Paper (Held On 19 May 2012)
A)
\[[PtC{{l}_{2}}{{(N{{H}_{3}})}_{2}}]\]
done
clear
B)
\[{{[NiC{{l}_{4}}]}^{2-}}\]
done
clear
C)
\[MnO_{4}^{-}\]
done
clear
D)
\[CrO_{4}^{2-}\]
done
clear
View Answer play_arrow
-
question_answer30) Reagent used to convert allyl alcohol to acrole in is
JEE Main Online Paper (Held On 19 May 2012)
A)
\[Mn{{O}_{2}}\]
done
clear
B)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
C)
\[Os{{O}_{4}}\]
done
clear
D)
\[KMn{{O}_{4}}\]
done
clear
View Answer play_arrow
 is
JEE Main Online Paper (Held On 19 May 2012)
is
JEE Main Online Paper (Held On 19 May 2012)
 JEE Main Online Paper (Held On 19 May 2012)
JEE Main Online Paper (Held On 19 May 2012)
 JEE Main Online Paper (Held On 19 May 2012)
JEE Main Online Paper (Held On 19 May 2012)