A) ethene
B) propene
C) butene-1
D) butene-2
Correct Answer: B
Solution :
(i) \[\underset{propene}{\mathop{C{{H}_{3}}CH}}\,=C{{H}_{2}}+{{O}_{3}}\xrightarrow{{}}\]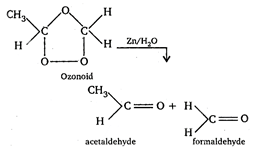 (ii) Ethene gives two molecules of formaldehyde on ozonolysis. (iii) Butene-1 gives one molecule of formaldehyde and one molecule of propanaldehyde on ozonolysis. (iv) Butene-2 gives two molecules of ethanol on ozonolysis.
(ii) Ethene gives two molecules of formaldehyde on ozonolysis. (iii) Butene-1 gives one molecule of formaldehyde and one molecule of propanaldehyde on ozonolysis. (iv) Butene-2 gives two molecules of ethanol on ozonolysis.
You need to login to perform this action.
You will be redirected in
3 sec
