A) \[C{{H}_{3}}COCl\xrightarrow{LiAl{{H}_{4}}}\]
B) \[{{C}_{2}}{{H}_{5}}CHO\xrightarrow[{{H}^{+}}/{{H}_{2}}O]{C{{H}_{3}}MgBr}\]
C) \[{{(C{{H}_{3}})}_{2}}CH{{C}_{2}}{{H}_{5}}\xrightarrow{Cu}\]
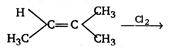
D)
Correct Answer: B
Solution :
Key Idea: The chiral carbon atom has 4 different groups or atoms attached to it. \[\underset{propanal}{\mathop{{{C}_{2}}{{H}_{5}}CHO}}\,+\underset{Grignard\text{ }reagent}{\mathop{C{{H}_{3}}MgBr}}\,\xrightarrow{{}}\]\[{{C}_{2}}{{H}_{5}}-\overset{\begin{smallmatrix} OMgBr \\ | \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{3}}\] \[{{\downarrow }^{{{H}_{2}}O/{{H}^{+}}}}\] \[{{C}_{2}}{{H}_{5}}-\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\mathop{C}}\,}}\,-C{{H}_{3}}\] \[{{C}^{*}}\] is asymmetric carbon atom.You need to login to perform this action.
You will be redirected in
3 sec
