A) \[C{{H}_{3}}-\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,}}\,-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\]
B) \[C{{H}_{3}}-C{{H}_{2}}-\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,}}\,-C{{H}_{2}}-C{{H}_{3}}\]
C) \[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{CH}}\,}}\,\]
D) \[C{{H}_{3}}-\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{C}}\,H-\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{C}}\,H-C{{H}_{2}}-C{{H}_{3}}\]
Correct Answer: A
Solution :
(i)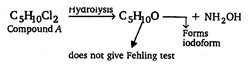 \[{{C}_{5}}{{H}_{10}}O\] must be aldehyde or ketone because it reacts with \[N{{H}_{2}}OH\]. (ii) It must be aldehyde as it does not give Fehling test. (iii) It must have \[C{{H}_{2}}CO\] group because it gives iodoform test. \[{{H}_{3}}C-\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,}}\,-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\xrightarrow{hydrolysis}\] \[\underset{(A)}{\mathop{{{C}_{5}}{{H}_{10}}C{{l}_{2}}}}\,\]
\[{{C}_{5}}{{H}_{10}}O\] must be aldehyde or ketone because it reacts with \[N{{H}_{2}}OH\]. (ii) It must be aldehyde as it does not give Fehling test. (iii) It must have \[C{{H}_{2}}CO\] group because it gives iodoform test. \[{{H}_{3}}C-\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,}}\,-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\xrightarrow{hydrolysis}\] \[\underset{(A)}{\mathop{{{C}_{5}}{{H}_{10}}C{{l}_{2}}}}\,\] 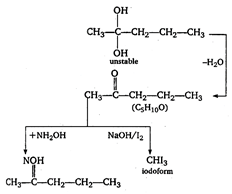
You need to login to perform this action.
You will be redirected in
3 sec
