A)
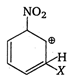
B)

C)
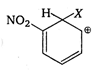
D)
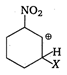
Correct Answer: A
Solution :
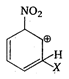 is most stable because electron attracting group (-I group) \[-N{{O}_{2}}\] is attached to it and positive charge of arenium ion do not come to fie carbon directly attached to \[N{{O}_{2}}\] group.
is most stable because electron attracting group (-I group) \[-N{{O}_{2}}\] is attached to it and positive charge of arenium ion do not come to fie carbon directly attached to \[N{{O}_{2}}\] group.
You need to login to perform this action.
You will be redirected in
3 sec
