A) more, ortho/para
B) less, ortho/para
C) more, meta
D) less, meta
Correct Answer: B
Solution :
-Cl group (-7) of chlorobenzene being deactivating, deactivates the benzene nucleus towards electrophilic substitution. Hence, chlorobenzene is less reactive as compared to benzene towards electrophilic substitution. Moreover chlorobenzene has the following canonical forms: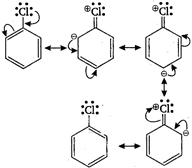 From the above, it is clear that Cl-atom increases electron density at ortho and para-position. Thus, it directs the incoming electrophile to the ortho/para position.
From the above, it is clear that Cl-atom increases electron density at ortho and para-position. Thus, it directs the incoming electrophile to the ortho/para position. 
You need to login to perform this action.
You will be redirected in
3 sec
