A) \[ds{{p}^{3}}\]
B) \[s{{p}^{3}}{{d}^{2}}\]
C) \[s{{p}^{3}}\]
D) \[{{d}^{2}}s{{p}^{3}}\]
Correct Answer: D
Solution :
In \[{{(Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\], Co is present as \[C{{o}^{3+}}\]. \[_{27}CO=[Ar]3{{d}^{7}}4{{s}^{2}}\]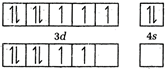 \[C{{O}^{3+}}=[Ar]\] \[{{[CO{{(N{{H}_{3}})}_{6}}]}^{3+}}=[Ar]\]
\[C{{O}^{3+}}=[Ar]\] \[{{[CO{{(N{{H}_{3}})}_{6}}]}^{3+}}=[Ar]\] 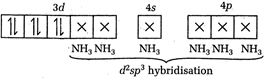
You need to login to perform this action.
You will be redirected in
3 sec
