A) \[{{d}^{10}}\]
B) \[{{d}^{8}}\]
C) \[{{d}^{6}}\]
D) \[{{d}^{4}}\]
Correct Answer: A
Solution :
When the configuration is d10, the distribution of electrons in the presence of either strong or weak field ligands is shown as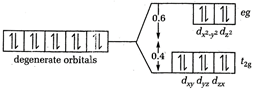 The electrons entering in the \[{{t}_{2g}}\] set stabilize the complex ion by \[0.4\,\Delta \] units while that entering in higher energy, \[{{e}_{g}}\] set, destabilise the complex ion by \[0.6\,\Delta \] units. Thus, \[CFSE=(0.4\times 6)-(0.6\times 4)=0\]
The electrons entering in the \[{{t}_{2g}}\] set stabilize the complex ion by \[0.4\,\Delta \] units while that entering in higher energy, \[{{e}_{g}}\] set, destabilise the complex ion by \[0.6\,\Delta \] units. Thus, \[CFSE=(0.4\times 6)-(0.6\times 4)=0\]
You need to login to perform this action.
You will be redirected in
3 sec
