A) \[{{d}^{2}}s{{p}^{3}}\]
B) \[s{{p}^{3}}\]
C) \[ds{{p}^{3}}\]
D) \[ds{{p}^{2}}\]
Correct Answer: C
Solution :
\[_{26}Fe=[Ar]\,3{{d}^{6}}4{{s}^{2}}\] In \[Fe{{(CO)}_{5}}\], CO being strong field ligand, causes pairing. Thus, Fe in Fe \[{{(CO)}_{5}}=[Ar]\]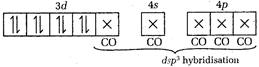 Thus, CO overlaps with the \[ds{{p}^{3}}\]hybrid orbitals of Fe atom.
Thus, CO overlaps with the \[ds{{p}^{3}}\]hybrid orbitals of Fe atom.
You need to login to perform this action.
You will be redirected in
3 sec
