A) The \[{{S}_{8}}\] ring is not planar
B) Oxygen is more electronegative than sulphur
C) \[S{{F}_{4}}\] exists, but \[O{{F}_{4}}\] does not exist
D) \[S{{O}_{3}}\] and \[SO_{3}^{2-}\] both have trigonal planar geometry
Correct Answer: D
Solution :
\[S{{O}_{3}}\] is trigonal planar due to \[s{{p}^{2}}\]hybridised sulphur.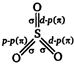 \[SO_{3}^{2-}\] is \[s{{p}^{3}}\] hybridised but pyramidal due to lone pair of electrons.
\[SO_{3}^{2-}\] is \[s{{p}^{3}}\] hybridised but pyramidal due to lone pair of electrons. 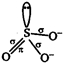
You need to login to perform this action.
You will be redirected in
3 sec
