A) o-nitrobenzoic acid is stronger than 3, 5 dinitrobenzoic acid in \[{{H}_{2}}O\]
B) Branched carboxylic acids are more acidic than unbranched acids
C)
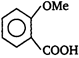 is stronger acid than
is stronger acid than 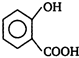
D) Butanoic acid is stronger acid than succinic acid
Correct Answer: A
Solution :
[a] o-nitrobenzoic acid is the strongest acid among all the nitrobenzoic acids due to ortho effect and stabilization of carboxylate ion by nitro group.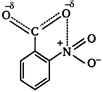 [b] Branched carboxylic acids are less acidic than unbranched acids due to +\[I\]-effect of alkyl groups. [c] o-hydroxybenzoic acid is stronger acid than o-methoxybenzoic acid due to stabilization of carboxylate ion by H-bonding.
[b] Branched carboxylic acids are less acidic than unbranched acids due to +\[I\]-effect of alkyl groups. [c] o-hydroxybenzoic acid is stronger acid than o-methoxybenzoic acid due to stabilization of carboxylate ion by H-bonding. 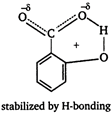
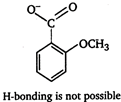 [d] Succinic acid is stronger acid than butanoic acid. This is due to -I-effect of one carboxyl group on the other end.
[d] Succinic acid is stronger acid than butanoic acid. This is due to -I-effect of one carboxyl group on the other end.
You need to login to perform this action.
You will be redirected in
3 sec
