A) bromine, 2-bromo aniline
B) bromine, 2,4,6 tribromoaniline
C) chloroform \[\text{ }\!\!|\!\!\text{ }\,\text{KOH,}\] phenyl cyanide
D) acetyl chloride, benzanilide
Correct Answer: B
Solution :
Aniline reacts with \[\text{B}{{\text{r}}_{2}}\]to give 2,4,6- tribromoaniline not bromoaniline as :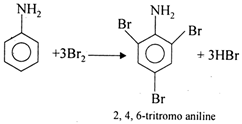
You need to login to perform this action.
You will be redirected in
3 sec
