A) I, II, III
B) II, III, I
C) II, I, III
D) I, III, II
Correct Answer: B
Solution :
Structure of \[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{3}}}\]is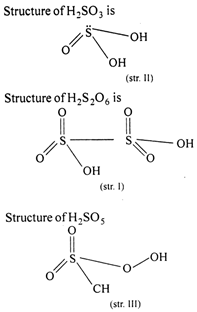 So, the correct order of increasing number of bonds is II, III, I
So, the correct order of increasing number of bonds is II, III, I
You need to login to perform this action.
You will be redirected in
3 sec
