| List 1 | List 2 | ||
| A | Alkanes | 1. | Molasses |
| B | Ethers | 2. | Hell-Volhard-Zelinsky reaction |
| C | \[\alpha -\] halogenation | 3. | Williamson synthesis |
| D | Ethyl alcohol | 4. | Wurtz reaction |
| 5. | Aldol condensation |
A) A-4 B-3 C-2 D-1
B) A-3 B-4 C-2 D-5
C) A-2 B-5 C-3 D-4
D) A-5 B-2 C-3 D-4
Correct Answer: A
Solution :
Wurtz reaction: Alkyl halide when heated with metallic sodium and zinc powder in ether gives alkane by joining two alkyl groups. \[2RX+2Na\xrightarrow{\text{ether}}R.R+2NaX\] Williamson synthesis: It is the laboratory preparation of ether. \[\underset{\text{Sod}\text{.alkoxide}}{\mathop{RONa}}\,+\underset{\text{Alkyl}}{\mathop{XR}}\,\xrightarrow{\Delta }\underset{\text{Ether}}{\mathop{ROR}}\,+NaX\] Hell-Volhard-Zelinsky reaction: \[\alpha -\] chloro or \[\alpha -\] bromo acids are usually prepared by first converting the acid into its chloride and bromide and then carrying out chlorination or bromination. Thus is called HV Z reaction. So, is for \[\alpha -\] halogenation. \[\underset{\text{Add}}{\mathop{RC{{H}_{2}}COOH}}\,\xrightarrow{\text{P + B}{{\text{r}}_{\text{2}}}}\underset{\text{Acid}\,\text{bromide}}{\mathop{RC{{H}_{2}}CO.Br}}\,\]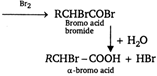 Aldol condensation: When aldehydes and ketones having \[\alpha -\] hydogen atoms are treated with alkali they undergo self-addition between two molecules to form a \[\beta -\] hydroxy aldehyde or ketone (known as aldol). This addition is usually followed by elimination of a water molecule resulting in overall condensation reaction. \[C{{H}_{3}}CHO+C{{H}_{3}}CHO\xrightarrow{O{{H}^{-}}}\underset{\begin{smallmatrix} \beta -\text{hydroxy}\,\text{butyraldehyde} \\ \text{(Aldol)} \end{smallmatrix}}{\mathop{C{{H}_{3}}-\underset{OH}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{2}}CHO}}\,\]Molasses: Ethyl alcohol is obtained from molasses. It is obtained as a byproduct of sugarindustry.
Aldol condensation: When aldehydes and ketones having \[\alpha -\] hydogen atoms are treated with alkali they undergo self-addition between two molecules to form a \[\beta -\] hydroxy aldehyde or ketone (known as aldol). This addition is usually followed by elimination of a water molecule resulting in overall condensation reaction. \[C{{H}_{3}}CHO+C{{H}_{3}}CHO\xrightarrow{O{{H}^{-}}}\underset{\begin{smallmatrix} \beta -\text{hydroxy}\,\text{butyraldehyde} \\ \text{(Aldol)} \end{smallmatrix}}{\mathop{C{{H}_{3}}-\underset{OH}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{2}}CHO}}\,\]Molasses: Ethyl alcohol is obtained from molasses. It is obtained as a byproduct of sugarindustry.
You need to login to perform this action.
You will be redirected in
3 sec
