A) A and D are isotopes
B) B and C are isodiaphers
C) C and D are isobars
D) A and B are isotopes
Correct Answer: D
Solution :
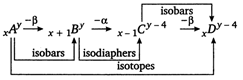 In the above process A and B are not isotopes as isotopes have same atomic number but different mass number.
In the above process A and B are not isotopes as isotopes have same atomic number but different mass number.
You need to login to perform this action.
You will be redirected in
3 sec
