A) Both (A) and (R) are true and (R) is the correct explanation of
B) Both (A) and (R) are true but (R) is not the correct explanation of
C) is true but (R) is not true
D) is not true but (R) is true
Correct Answer: B
Solution :
According to Lowry-Bronsted theory, acids have a tendency to give proton and bases have a tendency to accept proton. A single substance can give as well as accept proton, thus car behave like both eg,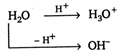 (He called such substance amphoteric.)\[\text{Acid + base}\xrightarrow{{}}\text{salt}\] Thus, both Assertion and Reason are correct but reason is not the correct explanation of Assertion.
(He called such substance amphoteric.)\[\text{Acid + base}\xrightarrow{{}}\text{salt}\] Thus, both Assertion and Reason are correct but reason is not the correct explanation of Assertion.
You need to login to perform this action.
You will be redirected in
3 sec
