A) m-bromonitrobenzene
B) o-bromonitrobenzene
C) p-bromonitrobenzene
D) Mixture of o - and p -bromonitrobenzene
Correct Answer: A
Solution :
\[-\text{N}{{\text{O}}_{\text{2}}}\]group being electron withdrawing decreases the electron density at ortho/para position. Thus, now electrophile attacks at meta position as it is electron rich as compared to ortho/para positions. \[B{{r}_{2}}+F{{e}^{3+}}\xrightarrow[{}]{{}}F{{e}^{3+}}+\underset{\text{electrophile}}{\mathop{B{{r}^{+}}}}\,\]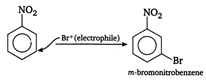
You need to login to perform this action.
You will be redirected in
3 sec
