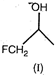
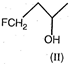
A) \[I>II>III>IV\]
B) \[I>III>II>IV\]
C) \[IV>III>II>I\]
D) \[IV>I>II>III\]
Correct Answer: C
Solution :
This is nucleophilic substitution reaction, hence stabler the carbocation, more reactive will be the alcohol. The order of stability of carbocations is: \[Ph-\overset{+}{\mathop{C}}\,{{H}_{2}}>C{{H}_{3}}-\overset{+}{\mathop{C}}\,H-C{{H}_{3}}\] \[>FC{{H}_{2}}-C{{H}_{2}}-\overset{+}{\mathop{C}}\,H-C{{H}_{3}}\] \[>FC{{H}_{2}}-\overset{+}{\mathop{C}}\,H-C{{H}_{3}}\]You need to login to perform this action.
You will be redirected in
3 sec
