A) \[{{H}_{2}}O\]
B) \[{{H}_{2}}S\]
C) \[{{H}_{2}}Se\]
D) \[{{H}_{2}}Te\]
Correct Answer: A
Solution :
Water \[({{H}_{2}}O)\] is least volatile due to the association of \[{{H}_{2}}O\] molecules through hydrogen bonding. However hydrogen bonding is not present in other hydrides.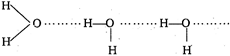
You need to login to perform this action.
You will be redirected in
3 sec
