A) \[Ca{{(OH)}_{2}}+2HCl\xrightarrow{{}}CaC{{l}_{2}}+2{{H}_{2}}O\]
B) \[2{{S}_{2}}O_{7}^{2-}+2{{H}_{2}}O\xrightarrow{{}}2SO_{4}^{2-}+4{{H}^{+}}\]
C) \[BaC{{l}_{2}}+MgS{{O}_{4}}\xrightarrow{{}}BaS{{O}_{4}}+MgC{{l}_{2}}\]
D) \[C{{u}_{2}}S+2FeO\xrightarrow{{}}2Cu+2Fe+S{{O}_{2}}\]
Correct Answer: D
Solution :
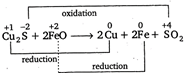 In this reaction Cu and Fe undergo reduction while sulphur undergoes oxidation. Hence, this is a redox reaction.
In this reaction Cu and Fe undergo reduction while sulphur undergoes oxidation. Hence, this is a redox reaction.
You need to login to perform this action.
You will be redirected in
3 sec
