A) Chlorobenzene
B) Tert-butylchloride
C) Isopropyl chloride
D) None of the above
Correct Answer: A
Solution :
\[C-Cl\]bond in aryl chloride is stable due to delocalisation of electron by resonance. Also \[CCl\]bond possess a double bond character like vinyl chloride, hence \[{{S}_{N}}\]reactions are not possible in chlorobenzene under ordinary conditions.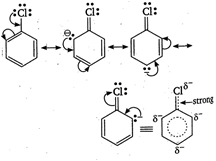
You need to login to perform this action.
You will be redirected in
3 sec
