A) \[N{{H}_{3}}\]
B) \[~{{H}_{2}}O\]
C) \[~B{{F}_{3}}\]
D) \[~C{{O}_{2}}\]
Correct Answer: A
Solution :
\[\text{N}{{\text{H}}_{\text{3}}}\]molecule in its valence shell has three bond pairs of electrons and one lone pair of electrons. The shape of \[\text{N}{{\text{H}}_{\text{3}}}\]molecule is pyramidal due to the presence of one lone pair electron. It has \[s{{p}^{3}}\]hybridisation.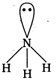
You need to login to perform this action.
You will be redirected in
3 sec
