A) \[Cl{{F}_{3}}\]
B) \[N{{H}_{3}}\]
C) \[B{{F}_{3}}\]
D) \[{{H}_{2}}O\]
Correct Answer: A
Solution :
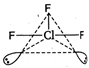 \[Cl{{F}_{3}}\Rightarrow 3bp+2lp,\]\[ie,\]\[s{{p}^{3}}d\]hybridisation The shape of \[\text{Cl}{{\text{F}}_{\text{3}}}\] should be trigonal bipyramidal but presence of two lone pairs converts it into T-shape. (The shape of \[\text{N}{{\text{H}}_{\text{3}}}\]is pyramidal, \[{{\text{H}}_{\text{2}}}\text{O}\]is angular and that of \[\text{B}{{\text{F}}_{\text{3}}}\]is trigonal planar.)
\[Cl{{F}_{3}}\Rightarrow 3bp+2lp,\]\[ie,\]\[s{{p}^{3}}d\]hybridisation The shape of \[\text{Cl}{{\text{F}}_{\text{3}}}\] should be trigonal bipyramidal but presence of two lone pairs converts it into T-shape. (The shape of \[\text{N}{{\text{H}}_{\text{3}}}\]is pyramidal, \[{{\text{H}}_{\text{2}}}\text{O}\]is angular and that of \[\text{B}{{\text{F}}_{\text{3}}}\]is trigonal planar.)
You need to login to perform this action.
You will be redirected in
3 sec
