A) \[C{{H}_{3}}Cl\]
B) \[CHC{{l}_{3}}\]
C) \[C{{H}_{2}}C{{l}_{2}}\]
D) \[CC{{l}_{4}}\]
Correct Answer: D
Solution :
\[\text{CC}{{\text{l}}_{\text{4}}}\text{,}\] because of its symmetrical structure, has zero dipole moment.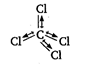 [Here, two dipoles vanish the effect of other two dipoles, thus the resultant dipole moment is zero.]
[Here, two dipoles vanish the effect of other two dipoles, thus the resultant dipole moment is zero.]
You need to login to perform this action.
You will be redirected in
3 sec
