A) \[{{[Ni{{(CN)}_{4}}]}^{2-}}\]
B) \[{{[NiC{{l}_{4}}]}^{2-}}\]
C) \[[Ni{{(CO)}_{4}}]\]
D) \[{{[CoC{{l}_{2}}{{(en)}_{2}}]}^{+}}\]
Correct Answer: B
Solution :
\[{{[NiC{{l}_{4}}]}^{2-}}\]has tetrahedral geometry due to \[s{{p}^{3}}\]hybridization. It contain two unpaired electron hence it is paramagnetic in nature.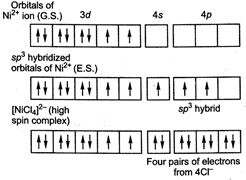
You need to login to perform this action.
You will be redirected in
3 sec
