A) for strong electrolytes
B) for weak electrolytes
C) both for strong and weak electrolytes
D) for non-electrolytes
E) None of these
Correct Answer: E
Solution :
Specific conductivity decreases with decrease in concentration for weak as well as strong electrolytes. It can shown as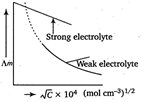 Molar conductivity however increases with decrease in concentration. Thus, the given question is incorrect.
Molar conductivity however increases with decrease in concentration. Thus, the given question is incorrect.
You need to login to perform this action.
You will be redirected in
3 sec
