A) their axes remain parallel to each other and perpendicular to the internuclear axis
B) their axes remain parallel to each other and parallel to the internuclear axis
C) their axes remain perpendicular to each other and parallel to the internuclear axis
D) their axes remain perpendicular to each other and perpendicular to the internuclear axis
Correct Answer: A
Solution :
A \[\pi -\]bond is formed by sidewise or parallel overlapping between atomic orbitals. During this their axes remain parallel to each other and perpendicular to the internuclear axis.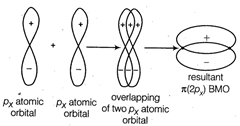
You need to login to perform this action.
You will be redirected in
3 sec
