A) 13.6 V
B) 3.4 V
C) 10.2 V
D) 1.89 V
Correct Answer: C
Solution :
From Bohrs concept, energy of atom is nth level is \[E=\frac{Rhc}{{{n}^{2}}}\]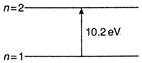 Energy required to raise an electron from ground state\[(n=1)\]to first excited state\[(n=2)\]is \[E={{E}_{2}}-{{E}_{1}}\] \[E=-3.4-(13.4)\] \[E=-3.4+13.4=10.2\,eV\]
Energy required to raise an electron from ground state\[(n=1)\]to first excited state\[(n=2)\]is \[E={{E}_{2}}-{{E}_{1}}\] \[E=-3.4-(13.4)\] \[E=-3.4+13.4=10.2\,eV\]
You need to login to perform this action.
You will be redirected in
3 sec
