A) \[Ni{{[{{({{H}_{2}}O)}_{6}}]}^{2+}}\]
B) \[{{K}_{2}}[NiC{{l}_{4}}]\]
C) \[{{K}_{2}}[Ni{{(CN)}_{4}}]\]
D) \[[Ni{{(CO)}_{4}}]\]
Correct Answer: D
Solution :
The oxidation number of Ni in\[[Ni{{(CO)}_{4}}]\]is zero.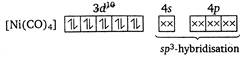 The hybridization in\[[Ni{{(CO)}_{4}}]\]is\[s{{p}^{3}}\]. Hence, the geometry of\[[Ni{{(CO)}_{4}}]\]is tetrahedral. It does not have any unpaired electrons.
The hybridization in\[[Ni{{(CO)}_{4}}]\]is\[s{{p}^{3}}\]. Hence, the geometry of\[[Ni{{(CO)}_{4}}]\]is tetrahedral. It does not have any unpaired electrons.
You need to login to perform this action.
You will be redirected in
3 sec
