A) \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
B) \[{{[Fe(C{{N}_{6}})]}^{3-}}\]
C) \[{{[Fe(C{{N}_{6}})]}^{4-}}\]
D) \[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}\]
Correct Answer: A
Solution :
Higher the number of unpaired electrons, higher is the magnetic moment.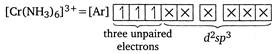 In\[{{[Fe{{(CN)}_{6}}]}^{3-}},\]one unpaired electron is present and in\[{{[Fe{{(CN)}_{6}}]}^{4-}}\]and\[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}\] both unpaired electrons are absent. Hence, \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]has the highest magnetic moment among the given complex ions. \[[\mu =\sqrt{3(3+2)}=3.878\,BM]\]. (\[C{{N}^{-}}\]is a strong field ligand, thus causes pairing.)
In\[{{[Fe{{(CN)}_{6}}]}^{3-}},\]one unpaired electron is present and in\[{{[Fe{{(CN)}_{6}}]}^{4-}}\]and\[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}\] both unpaired electrons are absent. Hence, \[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]has the highest magnetic moment among the given complex ions. \[[\mu =\sqrt{3(3+2)}=3.878\,BM]\]. (\[C{{N}^{-}}\]is a strong field ligand, thus causes pairing.)
You need to login to perform this action.
You will be redirected in
3 sec
