A) \[s{{p}^{3}}d\]
B) \[s{{p}^{3}}{{d}^{2}}\]
C) \[{{d}^{2}}s{{p}^{2}}\]
D) \[s{{p}^{3}}\]
Correct Answer: A
Solution :
In\[PC{{l}_{5}}\]five chlorine atoms directly attached to central atom and it has no lone pairs and no single electrons hence\[PC{{l}_{5}}\]has\[s{{p}^{3}}{{d}^{2}}\]hybridization.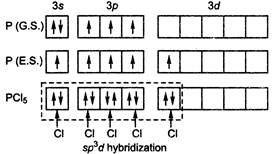
You need to login to perform this action.
You will be redirected in
3 sec
