A) dinitrogen monoxide and nitrogen monoxide
B) nitrogen monoxide and nitrogen dioxide
C) nitrogen dioxide and dinitrogen trioxide
D) dinitrogen trioxide and dinitrogen tetroxide
Correct Answer: B
Solution :
Nitrogen monoxide (nitric oxide) is paramagnetic in nature due to one unpaired electron (total 11 valence electrons). It can be represented by the following resonance structures \[\underset{\bullet \,\bullet }{\overset{\bullet }{\mathop{N}}}\,=\underset{\bullet \,\bullet }{\overset{\bullet \,\bullet }{\mathop{O}}}\,\overset{{}}{\longleftrightarrow}\underset{\bullet \,\bullet }{\overset{\bullet }{\mathop{^{\text{-O}}N}}}\,=\underset{\bullet \,\bullet }{\overset{\bullet \,\bullet }{\mathop{{{O}^{\oplus }}}}}\,\] Nitrogen dioxide is also paramagnetic due to one unpaired electron. Its structure is as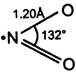
You need to login to perform this action.
You will be redirected in
3 sec
