A)
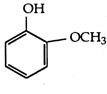
B)
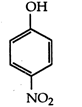
C) \[~\text{s}{{\text{p}}^{\text{2}}}\]
D) both (a) and (c)
Correct Answer: D
Solution :
Methoxy group, due to +1 effect, increase electron density on OH-group, thus making it less acidic. Thus, o-methoxy phenol and acetylene are less acidic than phenol, p-nitrophenol is more acidic than phenol.You need to login to perform this action.
You will be redirected in
3 sec
