A) \[NaHC{{O}_{3}}\]solution
B) \[5%\text{ }NaOH\]solution
C) neutral\[FeC{{l}_{3}}\]
D) bromine water
Correct Answer: A
Solution :
Key Idea:\[NaHC{{O}_{3}}\]reacts with\[-COOH\]group to give out\[C{{O}_{2}}\]while other groups do not react with it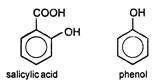 \[salicylic\text{ }acid+NaHC{{O}_{3}}\xrightarrow[{}]{{}}effervescence\] of \[C{{O}_{2}}\] \[Phenol+NaHC{{O}_{3}}\xrightarrow[{}]{{}}No\text{ }reaction\] \[\therefore \]\[NaHC{{O}_{3}}\]is used to distinguish between phenol and salicylic acid.
\[salicylic\text{ }acid+NaHC{{O}_{3}}\xrightarrow[{}]{{}}effervescence\] of \[C{{O}_{2}}\] \[Phenol+NaHC{{O}_{3}}\xrightarrow[{}]{{}}No\text{ }reaction\] \[\therefore \]\[NaHC{{O}_{3}}\]is used to distinguish between phenol and salicylic acid.
You need to login to perform this action.
You will be redirected in
3 sec
