A) 2-chlorobutane
B) 2,3-dichlorobufane
C) 2,3-dichloropentane
D) 2-hydroxypropanoic acid
Correct Answer: B
Solution :
(a) \[C{{H}_{3}}\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{\overset{*}{\mathop{C}}\,H}}\,C{{H}_{2}}C{{H}_{3}}\] One asymmetric carbon atom, forms\[d-\]and \[l-\]optical isomers. (b) Two asymmetric carbon atoms, forms \[d-,l-\]and meso forms.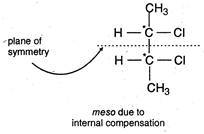 (c)\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{*}{\mathop{CH}}}\,-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{*}{\mathop{CH}}}\,-C{{H}_{2}}C{{H}_{3}}\] Two asymmetric carbon atoms but does not have symmetry. Hence, meso form is not formed. (d)\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\overset{*}{\mathop{CH}}}\,-COOH\] One asymmetric carbon atom, meso form is not formed.
(c)\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{*}{\mathop{CH}}}\,-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\overset{*}{\mathop{CH}}}\,-C{{H}_{2}}C{{H}_{3}}\] Two asymmetric carbon atoms but does not have symmetry. Hence, meso form is not formed. (d)\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\overset{*}{\mathop{CH}}}\,-COOH\] One asymmetric carbon atom, meso form is not formed.
You need to login to perform this action.
You will be redirected in
3 sec
