A) \[{{[Fe{{(CN)}_{6}}]}^{4-}}\]
B) \[{{[Mn{{(CN)}_{6}}]}^{4-}}\]
C) \[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
D) \[{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}\] (Atomic numbers\[Mn=25,\text{ }Fe=26,\text{ }Co=27,\] \[Ni=28\])
Correct Answer: D
Solution :
(a)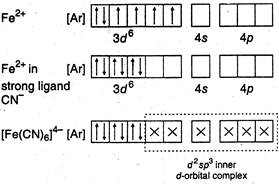 (b)
(b) 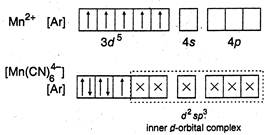 (c)
(c) 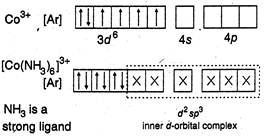 (d)
(d) 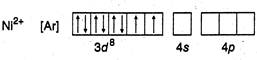 In this case also\[N{{H}_{3}}\]is a strong ligand but electrons remain unpaired since only one orbital is left vacant in\[3d\]. Thus
In this case also\[N{{H}_{3}}\]is a strong ligand but electrons remain unpaired since only one orbital is left vacant in\[3d\]. Thus 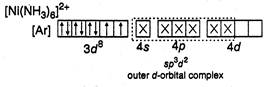
You need to login to perform this action.
You will be redirected in
3 sec
