A) acidic nature of\[{{H}_{2}}{{O}_{2}}\]
B) alkaline nature of\[{{H}_{2}}{{O}_{2}}\]
C) oxidising action of\[{{H}_{2}}{{O}_{2}}\]
D) reducing action of\[{{H}_{2}}{{O}_{2}}\]
Correct Answer: C
Solution :
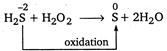 In this reaction, H^S is oxidised to S by\[{{H}_{2}}{{O}_{2}}\]so it represents oxidising action of\[{{H}_{2}}{{O}_{2}}\].
In this reaction, H^S is oxidised to S by\[{{H}_{2}}{{O}_{2}}\]so it represents oxidising action of\[{{H}_{2}}{{O}_{2}}\].
You need to login to perform this action.
You will be redirected in
3 sec
