A) \[{{C}_{6}}{{H}_{6}}+{{C}_{2}}{{H}_{5}}Cl\xrightarrow[{}]{AlC{{l}_{3}}}{{C}_{6}}{{H}_{5}}{{C}_{2}}{{H}_{5}}\]\[+HCl\]
B) \[{{C}_{2}}{{H}_{5}}OH+HCl\xrightarrow[{}]{ZnC{{l}_{2}}}{{C}_{2}}{{H}_{5}}Cl+{{H}_{2}}O\]
C) \[{{C}_{6}}{{H}_{5}}Cl+C{{H}_{3}}COCl\xrightarrow[{}]{AlC{{l}_{3}}}{{C}_{6}}{{H}_{5}}COC{{H}_{3}}\]\[+C{{l}_{2}}\]
D) \[{{C}_{6}}{{H}_{5}}Br+Mg\xrightarrow[{}]{Ether}{{C}_{2}}{{H}_{5}}MgBr\]
Correct Answer: A
Solution :
Alkyl substituted benzenes are prepared by the reaction between benzene and alkyl halide in the presence of a Lewis acid\[(AlC{{l}_{3}},\text{ }B{{F}_{3}},\text{ }FeC{{l}_{3}},\]etc.)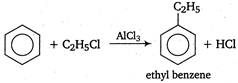 This reaction is known as Friedel-Crafts reaction.
This reaction is known as Friedel-Crafts reaction.
You need to login to perform this action.
You will be redirected in
3 sec
