A) \[ds{{p}^{2}},\]square planar, zero
B) \[ds{{p}^{2}},\]square planar, 1.73
C) \[s{{p}^{3}}{{d}^{2}},\]octahedral, zero
D) \[{{d}^{2}}s{{p}^{3}},\]octahedral, 1.73
Correct Answer: A
Solution :
\[{{[Ni{{(CN)}_{4}}]}^{2-}}\]ion has\[ds{{p}^{2}}\]hybridisation, zero magnetic moment and square planar structure.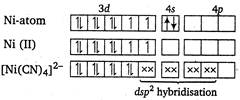 It has no unpaired electron hence, its magnetic moment is zero.
It has no unpaired electron hence, its magnetic moment is zero.
You need to login to perform this action.
You will be redirected in
3 sec
