A) \[{{V}_{2}}{{O}_{5}}\]
B) \[F{{e}_{2}}{{O}_{3}}\]
C) \[C{{r}_{2}}{{O}_{3}}\]
D) \[Cr{{O}_{3}}\]
Correct Answer: A
Solution :
Even at \[400-{{500}^{o}}C\] the rate of reaction is very low. Therefore, to increase the reaction velocity a suitable catalyst is used. Now a days most of the sulphuric acid plants use \[{{V}_{2}}{{O}_{5}}\] as a catalyst.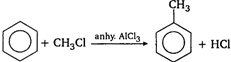
You need to login to perform this action.
You will be redirected in
3 sec
