A) different with \[1,\,\,\,0\] and \[2\] lone pairs of electrons on the central atoms, respectively
B) different with \[0,\,\,\,1\] and \[2\] lone pairs of electrons on the central atoms, respectively
C) the same with \[1,\,\,\,1\] and \[1\] lone pair of electrons on the central atoms, respectively
D) the-same with 2, 0 and 1 lone pairs of electrons on the central atoms, respectively
Correct Answer: A
Solution :
| Molecule | Structure | Hybridisation of central atom | Lone pair |
| \[S{{F}_{4}}\] | | \[s{{p}^{3}}d\] | one |
| \[Si{{F}_{4}}\] | 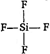 | \[s{{p}^{3}}\] | zero |
| \[ICI_{4}^{-}\] | | \[s{{p}^{3}}{{d}^{2}}\] | two |
You need to login to perform this action.
You will be redirected in
3 sec
