A) \[{{H}_{3}}P{{O}_{4}}\] is stronger acid, than\[{{H}_{2}}S{{O}_{3}}\]
B) In aqueous medium, \[HF\] is a stronger acid than\[HCl\]
C) \[HCl{{O}_{4}}\] is a weaker acid than\[HCl{{O}_{3}}\]
D) \[HN{{O}_{3}}\] is a stronger acid than\[HN{{O}_{2}}\]
Correct Answer: D
Solution :
Conjugate base of HNC^ is resonance stabilised than that of\[HN{{O}_{2}}\]. Hence, \[HN{{O}_{3}}\] is stronger acid than\[HN{{O}_{2}}\].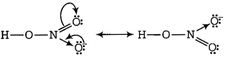
You need to login to perform this action.
You will be redirected in
3 sec
