A) Oxalic acid
B) Formic acid
C) Picric acid
D) Citric acid
Correct Answer: C
Solution :
Key Idea: Draw all the structures to find which of them has phenolic -OH group. [a] Oxalic acid \[\underset{COOH}{\overset{COOH}{\mathop{|}}}\,\] [b] From acid \[HCOOH\] [c] Picric acid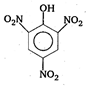 [d] Citric acid \[HOOC-C{{H}_{2}}-\underset{COOH}{\overset{OH}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-C{{H}_{2}}-COOH\] \[\therefore \]Picric acid has phenolic -OH group.
[d] Citric acid \[HOOC-C{{H}_{2}}-\underset{COOH}{\overset{OH}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-C{{H}_{2}}-COOH\] \[\therefore \]Picric acid has phenolic -OH group.
You need to login to perform this action.
You will be redirected in
3 sec
