A) 112
B) 512
C) 400
D) 614
Correct Answer: B
Solution :
Key Idea: The rate of reaction increases by \[{{(2)}^{n}}\]for every \[{{10}^{o}}\] rise in temperature.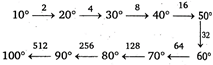 \[\therefore \]rate of reaction increase 512 times.
\[\therefore \]rate of reaction increase 512 times.
You need to login to perform this action.
You will be redirected in
3 sec
