A) (i) > (ii) > (iii) > (iv)
B) (ii) > (iv) > (iii) > (i)
C) (ii) > (iv) > (i) > (iii)
D) (ii) > (iii) > (iv) > (i)
Correct Answer: D
Solution :
(i)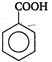 (ii)
(ii)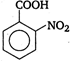 (iii)
(iii) 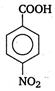 (iv)
(iv) 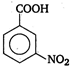 \[-\text{N}{{\text{O}}_{\text{2}}}\]group at any position shows electron withdrawing effect, thus acid strength is increased. But o-nitro benzoate ion is stabilised by intramolecular H-bonding like forces, hence its acid strength is maximum. Thus, the order of acid strength is (i) > (ii) > (iv) > (i).
\[-\text{N}{{\text{O}}_{\text{2}}}\]group at any position shows electron withdrawing effect, thus acid strength is increased. But o-nitro benzoate ion is stabilised by intramolecular H-bonding like forces, hence its acid strength is maximum. Thus, the order of acid strength is (i) > (ii) > (iv) > (i).
You need to login to perform this action.
You will be redirected in
3 sec
