A) colourless and diamagnetic
B) coloured and octahedral
C) colourless and paramagnetic
D) coloured and paramagnetic
Correct Answer: A
Solution :
In \[{{[Sc{{({{H}_{2}}O)}_{6}}]}^{3+}},\] oxidation state of Sc is \[+\,3.\]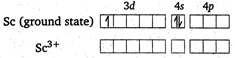 \[\because \] has no unpaired electrons; \[\therefore \]\[{{[Sc{{({{H}_{2}}O)}_{6}}]}^{3+}}\]is diamagnetic and colourless.
\[\because \] has no unpaired electrons; \[\therefore \]\[{{[Sc{{({{H}_{2}}O)}_{6}}]}^{3+}}\]is diamagnetic and colourless.
You need to login to perform this action.
You will be redirected in
3 sec
