A) \[O{{H}^{-}}\]
B) \[O{{R}^{-}}\]
C)
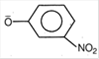
D)
![]()
Correct Answer: B
Solution :
Weakest acid has the strongest conjugate base. Since, R OH is the weakest acid, therefore \[\text{R}{{\text{O}}^{-}}\]is the strongest base.You need to login to perform this action.
You will be redirected in
3 sec
