A)

B)
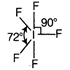
C)
D) None of these
Correct Answer: C
Solution :
Number of hybrid orbitals = number of \[bp+\]number of \[lp\] \[=5+1=6\] Thus, hybridisation is \[\text{s}{{\text{p}}^{\text{3}}}{{\text{d}}^{\text{2}}}\]but geometry due to the presence of one lone pair is square pyramidal.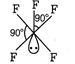
You need to login to perform this action.
You will be redirected in
3 sec
